.png)
Clinical Trial Management System (CTMS)
GAJA CTMS(Globally Accessible CTMS with Justified Accuracy) is specialized software used by pharmaceutical companies, CROs (Contract Research Organizations), research sites, and academic institutions to plan, track, and manage clinical trials more efficiently. It acts as a central hub for all trial-related operational, financial, and regulatory information.
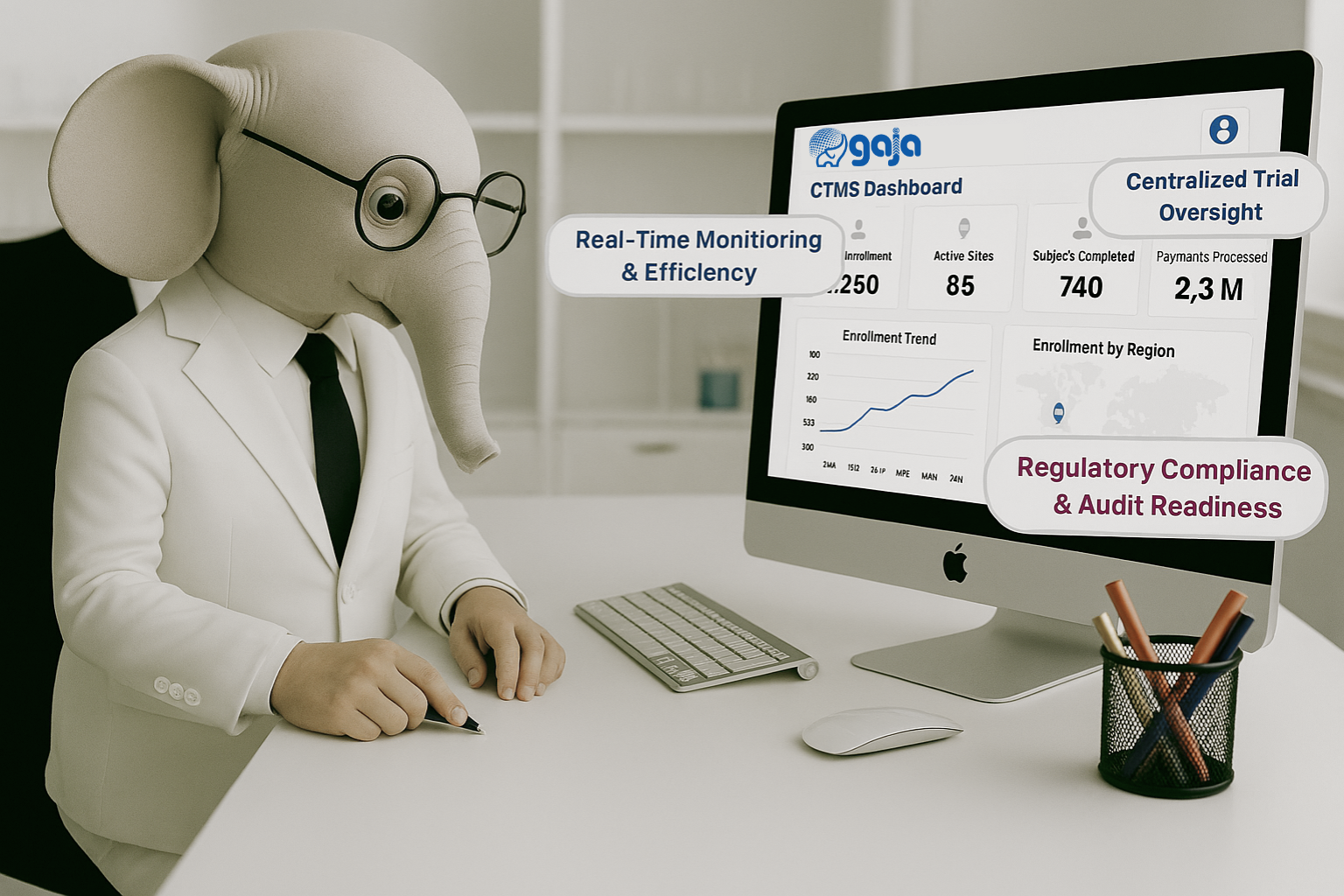
Why Choose GAJA CTMS for Your Clinical Trials?
- ✔ Centralized Trial Management All study, site, and subject information in one secure platform.
- ✔ Regulatory Compliance Supports ICH-GCP, FDA, and EMA standards with full audit trails.
- ✔ Financial Control Automates site payments, budgeting, and financial tracking.
- ✔ Real-Time Oversight Dashboards and reports for enrollment, milestones, and site performance.
- ✔ Seamless Integrations Works with EDC, eTMF, and RTSM for unified trial operations.
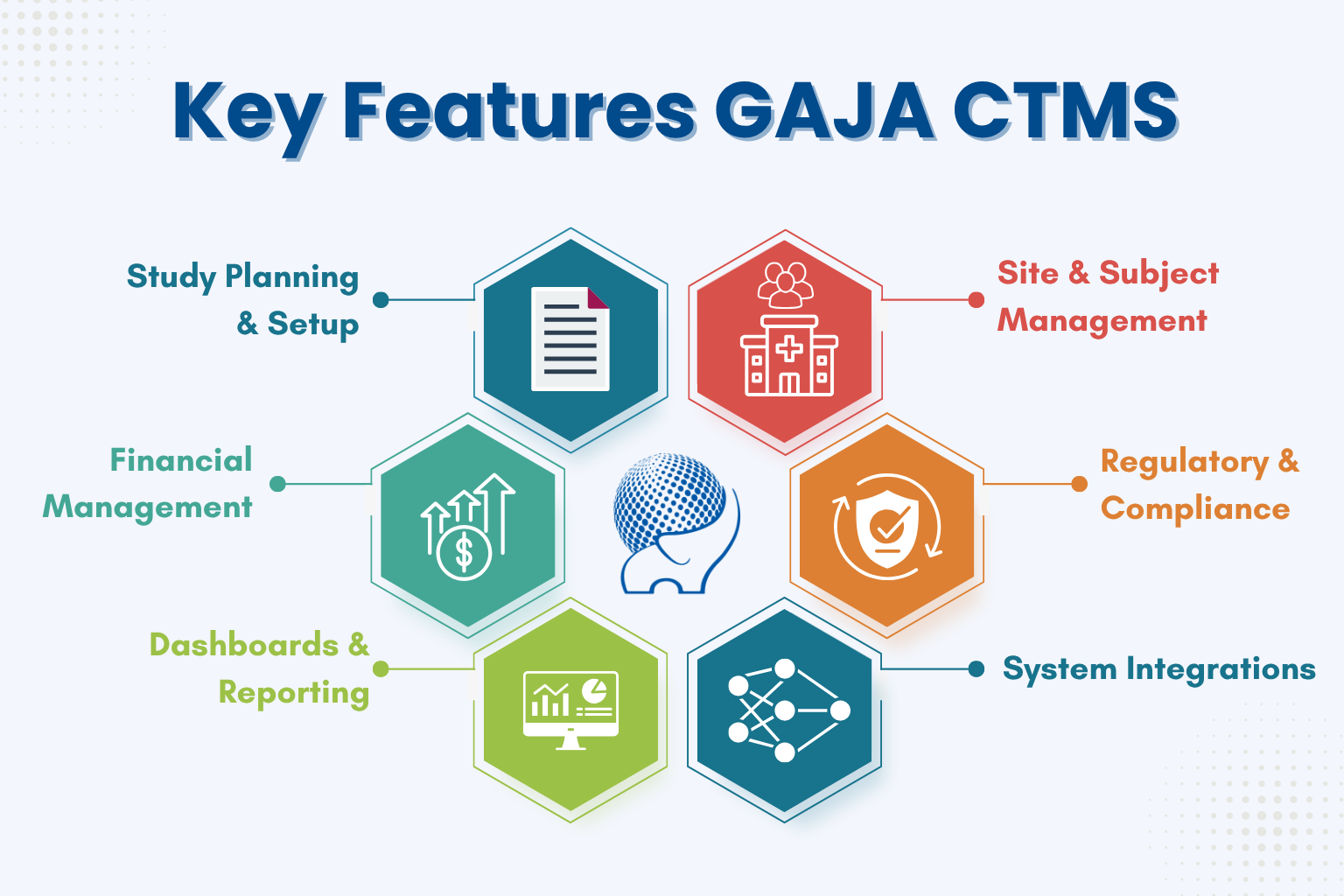
Features Of GAJA CTMS
- Study Planning & Setup Design protocols, plan timelines, and manage site selection in one place.
- Site & Subject Management Track enrollment, visits, and performance across all clinical sites.
- Financial Management Automate budgets, payments, and financial reconciliation with sites.
- Regulatory & Compliance Maintain audit trails and ensure compliance with GCP and regulatory bodies.
Benefits of GAJA CTMS
Operational Efficiency
Centralized system reduces duplication and manual work, improving productivity.
Regulatory Compliance
Helps maintain GCP, ICH, and FDA/EMA standards with built-in audit trails.
Real-Time Insights
Dashboards provide quick visibility into enrollment, site performance, and milestones.
Cost Control & Collaboration
Budgeting, forecasting, and financial tracking while enabling sites, sponsors, and CROs to work on one platform.
Who Uses CTMS?
Sponsors
Pharmaceutical, biotech, and medical device companies use CTMS to manage trials efficiently.
CROs
Contract Research Organizations manage multiple studies for sponsors using CTMS.
Academic Centers
Universities and research institutes leverage CTMS for clinical and investigator-led studies.
Clinical Sites
Hospitals and clinical sites use CTMS to track patients, visits, and compliance.
