.png)
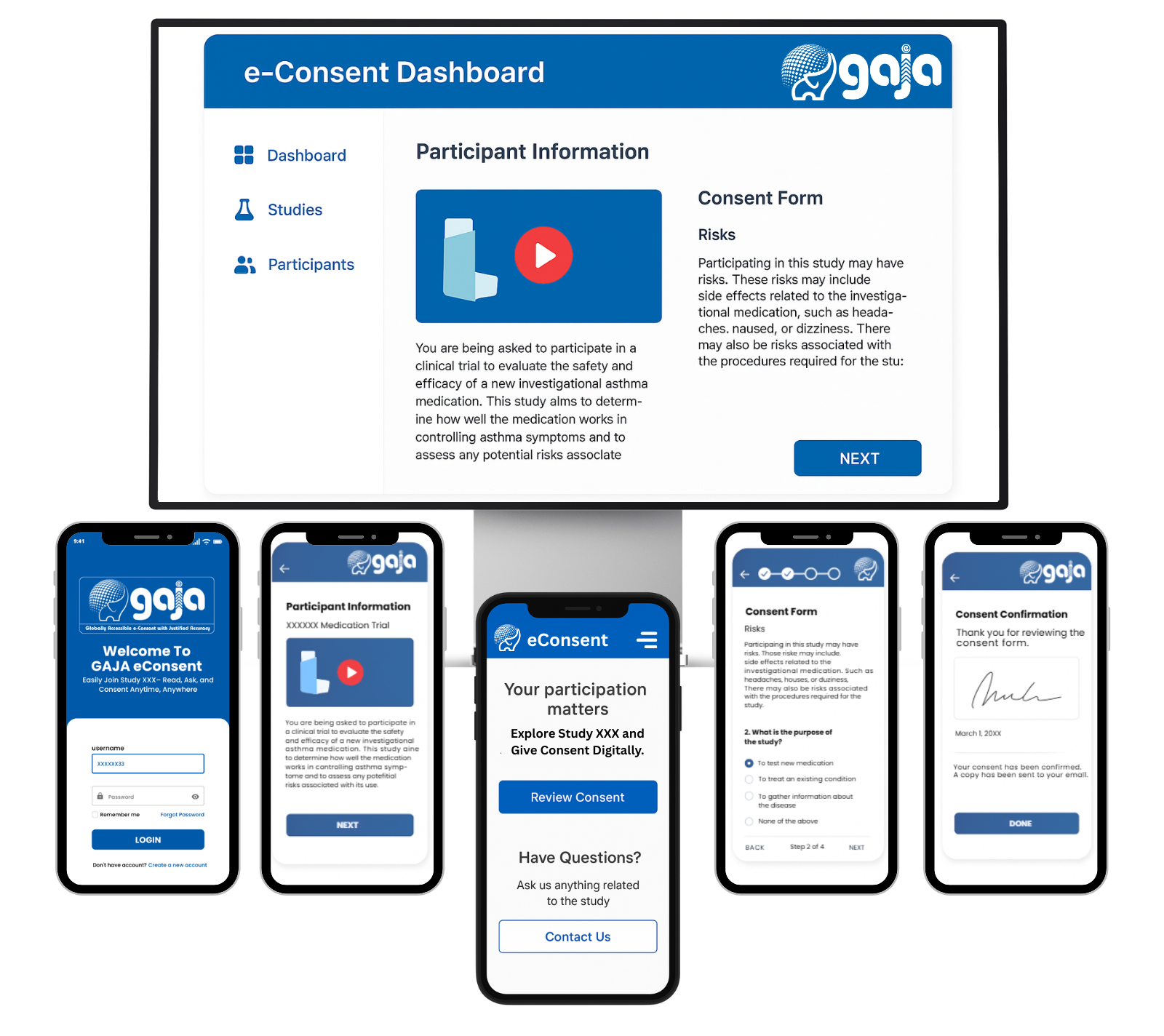
Electronic Informed Consent (e-Consent)
GAJA e-Consent(Globally Accessible e-Consent with Justified Accuracy) is a secure digital solution that enables participants to review, understand, and sign informed consent forms electronically.It simplifies the consent process, enhances patient comprehension, and ensures regulatory compliance in clinical trials.

Why Choose GAJA e-Consent for Your Clinical Trials?
- ✔ Enhanced Patient Understanding Multimedia (videos, images, FAQs) improves clarity and comprehension.
- ✔ Convenient & Remote Patients can review and sign consent forms anytime, anywhere.
- ✔ Regulatory Confidence Fully compliant with 21 CFR Part 11, ICH-GCP, and GDPR standards.
- ✔ Audit-Ready Tracks signatures, timestamps, and consent versions for transparency.
- ✔ Patient-Centric Provides interactive content in multiple languages for better accessibility.
- ✔ Integrated Workflow Seamlessly connects with GAJA EDC, eSource, and RTSM for end-to-end digital trial management.
Features Of GAJA e-Consent
- Interactive Content Videos, graphics, and quizzes to improve patient comprehension.
- Electronic Signatures Secure, compliant e-signature capture with timestamps.
- Version Control Automatic tracking of consent form updates and re-consents.
- Remote Access Patients and caregivers can review and sign forms online.
- Multi-Language Support Consent forms available in global languages.
- Audit Trails & Security Full traceability of consent activities.
Benefits of GAJA e-Consent
Improved Patient Understanding
Ensures error-free patient allocation and maintains study integrity.
Faster Enrollment
Tracks inventory in real time and predicts resupply needs to prevent shortages.
Enhanced Compliance
Provides instant visibility into enrollment, site activity, and supply status.
Regulatory Compliance
Fully adheres to 21 CFR Part 11 and ICH-GCP standards for secure, traceable operations.
